The Role of Incentives: How Policy Shapes Rare Disease Innovation
Developing treatments for rare diseases involves a unique combination of scientific, logistical, and economic challenges. While the need for therapies is clear, the path to development is often shaped by the realities of working with small and widely dispersed patient populations.
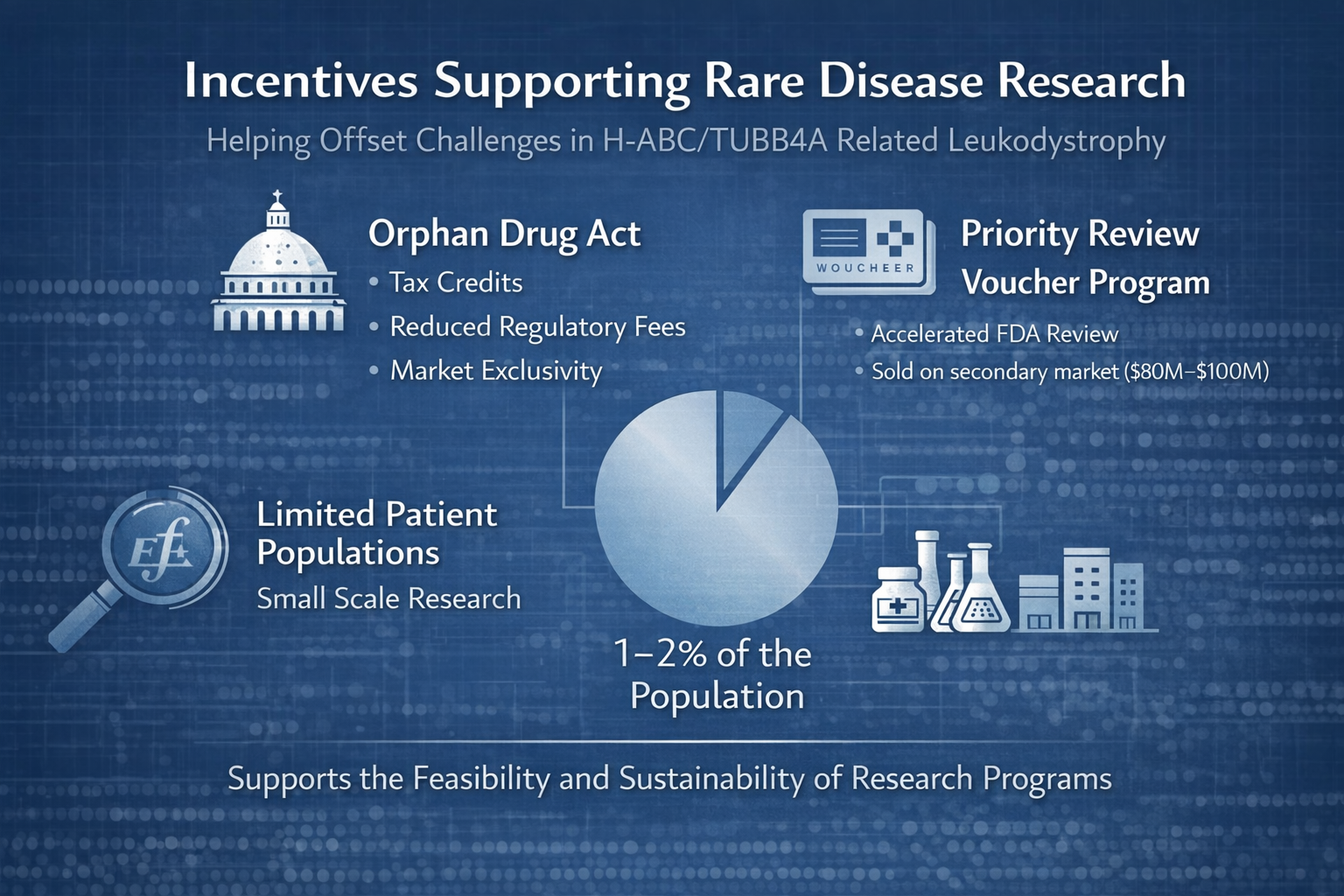
Bringing a new therapy from early research through regulatory approval can require substantial investment, with estimates often exceeding $1–2 billion. For conditions such as H-ABC/TUBB4A related leukodystrophy, where only a limited number of individuals have been identified worldwide, traditional market incentives alone are often insufficient to support this level of investment.
In response to these challenges, policy frameworks have been established to encourage research and development in rare diseases. The Orphan Drug Act created a structured approach to supporting therapies for conditions affecting small populations. Through programs administered by the U.S. Food and Drug Administration, developers may be eligible for incentives such as tax credits, reduced regulatory fees, and a period of market exclusivity following approval. These provisions are intended to help offset some of the financial risks associated with developing treatments for rare conditions.
Since the implementation of these policies, the number of therapies available for rare diseases has increased, with more than 600 orphan-designated drugs reaching the market. While many rare conditions still do not have approved treatments, this growth reflects a shift in how rare disease research is supported and prioritized.
Additional programs have been introduced to address specific gaps, particularly in pediatric rare diseases. The Rare Pediatric Disease Priority Review Voucherprogram provides an incentive for developing therapies for serious pediatric conditions. Under this program, a sponsor who receives approval for an eligible therapy may be awarded a voucher that allows for expedited review of another product. These vouchers can be transferred or sold, and in some cases have been valued in the range of $80 million to over $100 million, providing a mechanism to help offset development costs.
Even with these incentives, rare disease drug development remains complex. Clinical trials may involve small cohorts, limited natural history data, and participants located across multiple regions. These factors can make study design, recruitment, and data interpretation more challenging than in more common conditions.
For H-ABC/TUBB4A, these dynamics are particularly relevant. The small number of identified individuals, combined with variability in clinical presentation, underscores the importance of both scientific collaboration and supportive policy frameworks. Incentives do not eliminate the challenges, but they can influence whether research programs are initiated and sustained over time.
Policy discussions in this area continue to evolve, with ongoing consideration of how best to support innovation while maintaining accessibility and long-term sustainability. As research advances, the role of these programs remains closely tied to the ability to move potential therapies from concept to clinical evaluation.
For the H-ABC/TUBB4A community, these frameworks represent one component of a broader research environment. While they do not determine outcomes on their own, they help create conditions in which continued scientific progress is more achievable.