H-ABC / TUBB4A Leukodystrophy Community BLOG
The Hidden Costs of Rare Disease Caregiving
This blog focuses on the financial pressure many caregivers face while supporting someone living with H-ABC/TUBB4A related leukodystrophy or another rare neurological disease. Beyond medical care, families may carry the cost of travel to specialists, therapies, adaptive equipment, home modifications, medications, insurance deductibles, missed work, reduced hours, and daily caregiving needs that are not always covered by insurance. The central message is that caregivers should not have to navigate the financial side of rare disease alone, and asking for help is a practical step toward protecting both family stability and caregiver well-being.
Caregiver Support: Finding Help Before You Reach Empty
This blog is written for caregivers supporting someone living with H-ABC/TUBB4A related leukodystrophy or another rare neurological disease. It acknowledges the daily strain of appointments, paperwork, insurance issues, school needs, sleep disruption, financial pressure, and emotional exhaustion, while offering practical first steps for finding support.
Caregivers are encouraged to begin with one urgent need, ask the medical team for a social worker or care coordinator, keep key records in one place, seek respite before a crisis, and connect with therapy, school, insurance, disability, and rare disease resources. The central message is simple: caregivers are often the center of the care system, and they need support too.
From One Caregiver to Another: Lightening a Load That Never Fully Lifts
Rare disease caregiving is often a quiet, constant kind of work — filled with medical vigilance, disrupted sleep, therapy schedules, appointments, financial strain, emotional uncertainty, and isolation from everyday life.
For families affected by H-ABC/TUBB4A and other rare neurological diseases, the stress reaches beyond logistics; it can affect the body, mind, relationships, and overall well-being of both the person diagnosed and the caregiver.
This blog speaks from one caregiver to another, acknowledging the weight of that responsibility while offering practical, realistic ways to reduce the stress burden and remember that caregivers need care, support, and compassion too.
Building a Rare Disease Community: Why Connection Supports Progress
Connection within the rare disease community plays an important role in supporting both care and research, particularly for conditions such as H-ABC/TUBB4A related leukodystrophy where individuals may be geographically dispersed and limited in number. Advocacy organizations, including the Foundation to Fight H-ABC/TUBB4A, help facilitate these connections by sharing information, supporting participation in registries and studies, and fostering collaboration among families, clinicians, and researchers. Through these coordinated efforts, community engagement contributes to a broader understanding of the condition and helps support a more informed and connected approach to ongoing research and care.
Speech and Communication in H-ABC/TUBB4A Related Leukodystrophy: What We Know and What We’re Learning
Communication development in H-ABC/TUBB4A related leukodystrophy is influenced by changes in white matter that can affect motor control and the coordination required for speech. As a result, individuals may experience a range of communication differences, from early delays to changes over time, reflecting both the variability of the condition and its neurological basis. Approaches such as speech-language therapy and augmentative and alternative communication (AAC) can support functional communication, while ongoing research is increasingly recognizing communication as an important measure of daily function and quality of life.
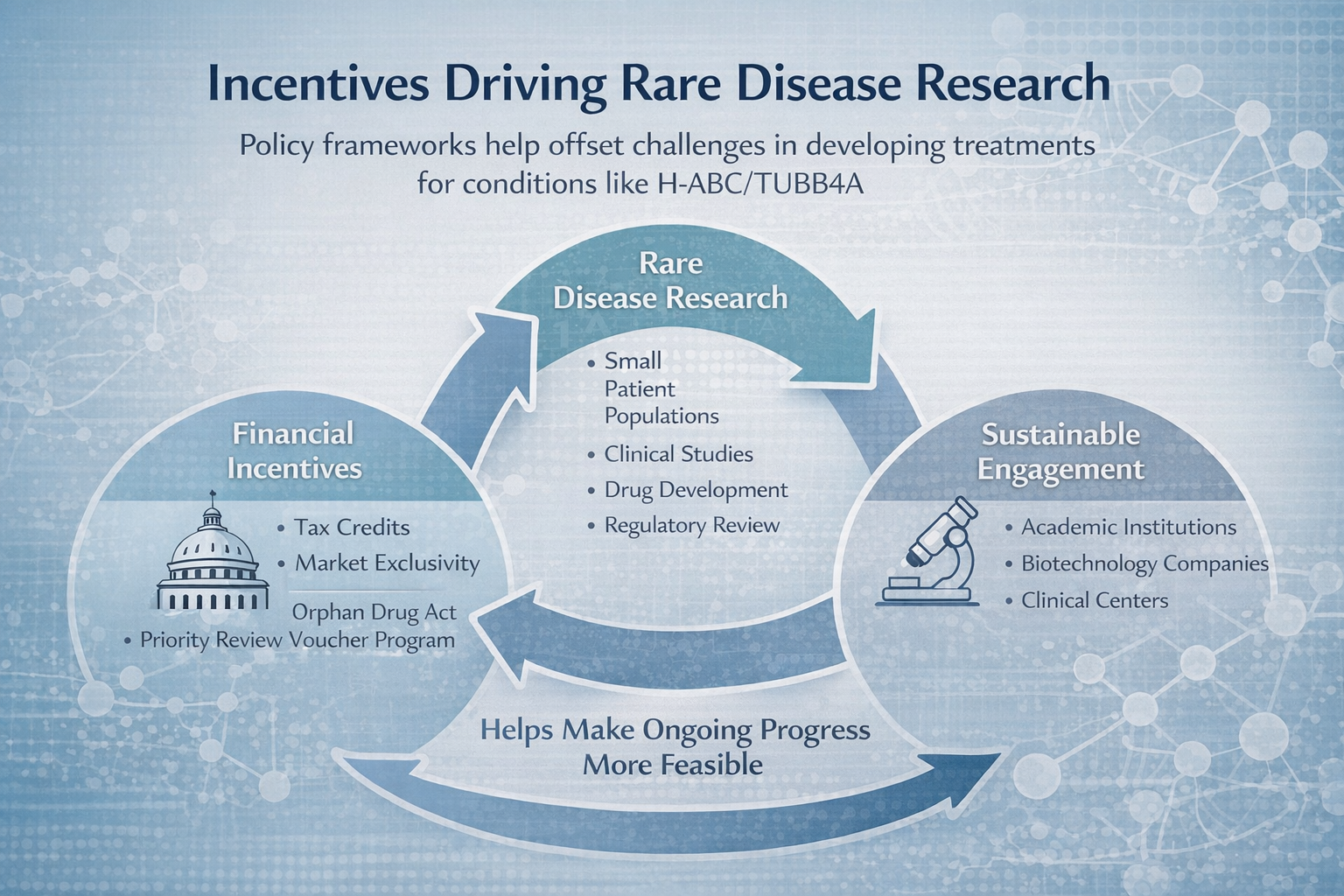
The Role of Incentives: How Policy Shapes Rare Disease Innovation
Policies such as the Orphan Drug Act and the Rare Pediatric Disease Priority Review Voucher program play an important role in supporting rare disease research by helping to offset the financial and logistical challenges associated with developing therapies for small patient populations. For conditions such as H-ABC/TUBB4A related leukodystrophy, where the number of identified individuals is limited, these incentives can influence whether research programs move forward and how resources are allocated. While they do not remove the complexities of drug development, they contribute to a framework that makes continued scientific progress more feasible and helps sustain engagement across research, clinical, and industry communities.
Natural History Studies: Why They Matter Before a Cure Exists
Natural history studies play an important role in rare disease research by helping to build a clear understanding of how a condition develops over time. In diseases such as H-ABC/TUBB4A related leukodystrophy, where relatively few individuals have been identified worldwide and clinical presentation can vary, this type of longitudinal observation provides essential context for interpreting change. By documenting how symptoms progress in the absence of treatment, natural history data helps inform clinical trial design, supports the identification of meaningful outcome measures, and contributes to a more structured path toward future therapeutic development.